![]() NOW ENROLLING
NOW ENROLLING
PDO Screen
Patient-derived colorectal cancer organoid screening platform
Translate your in vitro pharmacology data from your lab to the clinic
Despite recent advancements in colorectal cancer (CRC) treatment, the prognosis remains poor for patients with advanced metastatic disease.
Our Patient-Derived Organoid (PDO) Screen using CRC organoids allows you to accelerate your most promising clinical candidate or broaden the application of your FDA- and EMA-approved targeted therapy beyond initial indications.

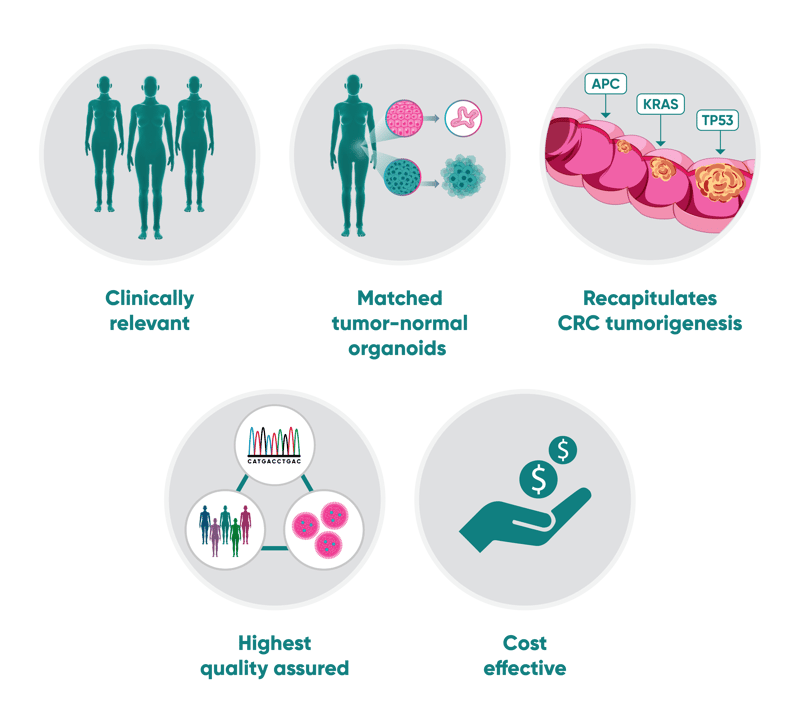
Benefit from:
- Predictivity: select your lead agent with the highest therapeutic potential and lowest toxicity thanks to testing simultaneously on tumor and normal patient-derived organoids
- Speed: get translatable data in a fraction of the time needed for an in vivo study
- Cost saving compared to stand-alone studies
- Patient relevance: HUB Organoids are patient-derived models that preserve the original tumor histo- and molecular pathology
- Quality assurance with frequent SNP checks and genomic characterizations
With years of experience in optimizing culture conditions, we have successfully established and maintained an extensive panel of patient-derived colorectal cancer organoids from primary and metastatic tumors that faithfully recapitulate the key genetic drivers of CRC progression. Additionally, our technology provides a unique framework to test off-target and off-tumor toxicities in patient-derived organoids from normal colorectal epithelia.
Screen features:
- ATP-based cell-viability assay with GR50 readouts
- 3D assay format with 96/384-well plate format
- Test up to 10 compounds in triplicates
- 10-point dose-response curve/drug
- 20 model screens: 14 CRC organoid lines capturing key cancer drivers, 6 normal organoid lines
- 3 matched tumor-normal organoid pairs to uncover off-tumor toxicities
- Representative of patient population heterogeneity with organoids derived from both primary and metastatic tumors
| MODEL ID | Cancer type | Mutated genes | Source | DNA seq | RNA seq | Matched normal organoid available |
|---|---|---|---|---|---|---|
| CRC-001 | Colorectal Cancer | BRAF, PIK3CA, RNF43 | Primary tumor | |||
| CRC-002 | Colorectal Cancer | KRAS, PIK3CA, TP53 | Primary tumor | |||
| CRC-003 | Colorectal Cancer | APC, KRAS | Primary tumor | |||
| CRC-004 | Colorectal Cancer | APC, KRAS, SOX9 | Primary tumor | |||
| CRC-005 | Colorectal Cancer | APC | Primary tumor | |||
| CRC-006 | Colorectal Cancer | APC, KRAS, TP53, SMAD4 | Primary tumor | |||
| CRC-007 | Colorectal Cancer | APC, KRAS, TP53, SMAD4 | Primary tumor | |||
| CRC-008 | Colorectal Cancer | APC, TP53 | Primary tumor | |||
| CRC-009 | Colorectal Cancer | APC, TP53 | Primary tumor | |||
| CRC-010 | Colorectal Cancer | APC, TP53, KRAS, EGFR | Primary tumor | |||
| CRC-011 | Colorectal Cancer | APC | Primary tumor | |||
| CRC-012 | Colorectal Cancer | APC, TP53, KRAS, SMAD4 | Metastatic Tumor | |||
| CRC-013 | Colorectal Cancer | APC, TP53, KRAS, EGFR | Metastatic Tumor | |||
| CRC-014 | Colorectal Cancer | APC, KRAS, SOX9 | Metastatic Tumor |
Forecast a champion for your preclinical drug development pipeline.
Limited slots available!

